
In the same way the anions can have lesser radius that the radius of its original neutral atom.The ionic radius is thus reduced than atomic radius.
However the nucleus attract the rest of the electrons more tightly towards the nucleus.

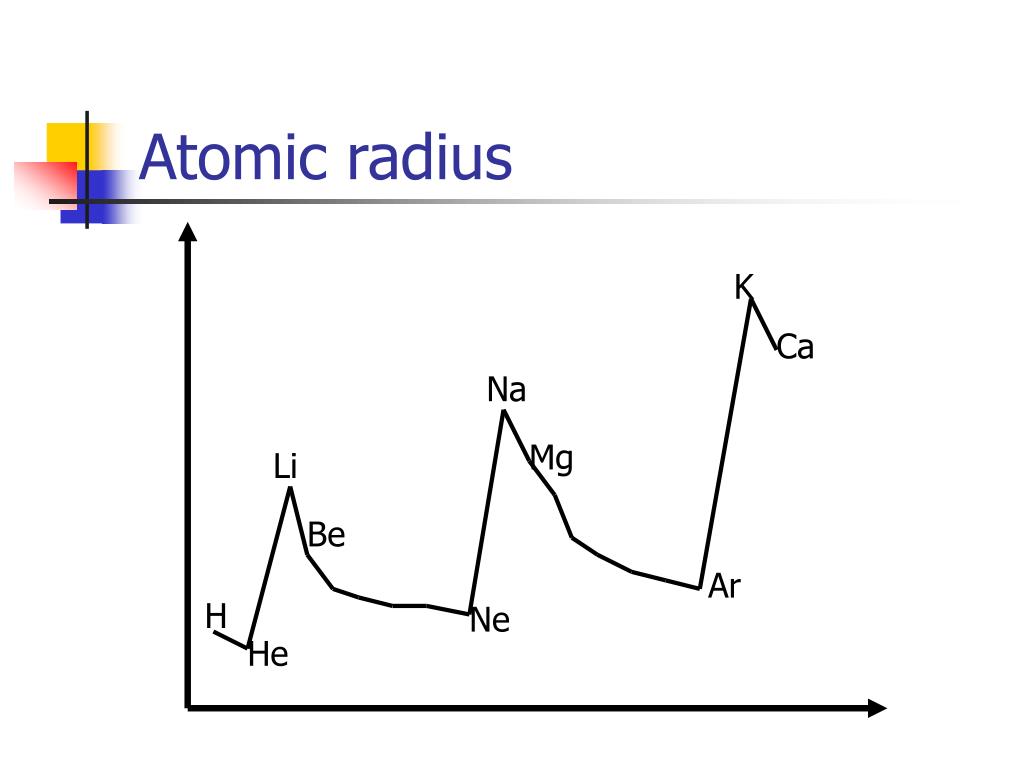
Thus sometime different methods are used to measure the radius while they are bonded in a molecule. As there are no physical existence of orbital in atoms, it is difficult to measure the atomic radius. And I think that your question initially meant to compare the size of #Al^(3+)# NOT #Al^(3-)# (largely unknown).Īnd now to the anions, and cations.and for a given PERIOD, a CATION should be smaller than an anion, inasmuch as we remove an electron, and the remaining electrons should be (and are) held more tightly at closer radii due to the absence of electronic shielding.The atomic radius is the size of the atom, typically measured by the distance from the nucleus of the atom to the electron clouds around the nucleus. For selenium, potassium, and bromine, we gots #n=4#.potassium should have the biggest atomic radius, followed by selenium, then bromine.i.e. Than 4th row potassium, selenium, and bromine.because its valence shell is #n=3# rather than #n=4#. And this is basic and assumed knowledge for 1st year.Īnd so we repair to the Table.and we compare, #Al#, #Se#, #K#, and #Br# ATOMS.Īluminum is a third-row element.and thus it SHOULD be smaller And of course with respect to a row, a Period of the Periodic Table as we face the Table, the size atoms (defined by their valence electronic radius) should DECREASE, and that with respect to a column, a Group of the Periodic Table, atomic radii should INCREASE down the Group. Reasonably, we address the radii of ions and atoms, by the radius of their VALENCE electron. ANIONS should be LARGER than their parent atoms?

